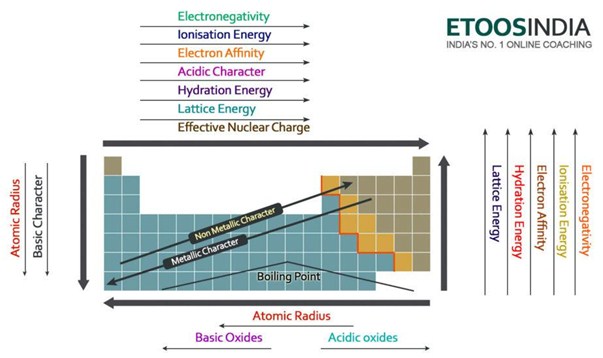 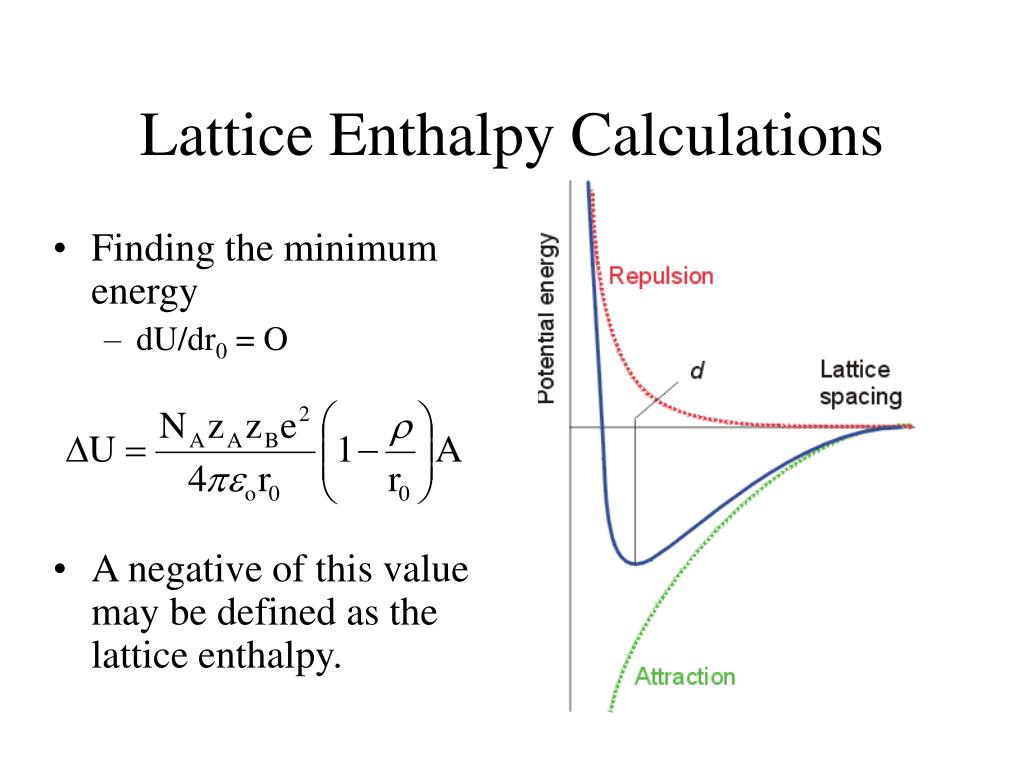
Therefore, the change in energy is always positive. The dissociation of a compound is always an endothermic process, meaning it will always require an input of energy. Dissociation energy is the energy required to break apart a compound.In general, electron affinity increases from left to right across the periodic table and decreases from top to bottom. Therefore, when used in calculating the lattice energy, we must remember to subtract the electron affinity, not add it. 
Usually, energy released would have a negative value, but due to the definition of electron affinity, it is written as a positive value in most tables. Electron Affinity is the energy released when an electron is added to a neutral atom or an ion.There are some excepts, usually due to the stability of half-filled and completely filled orbitals. 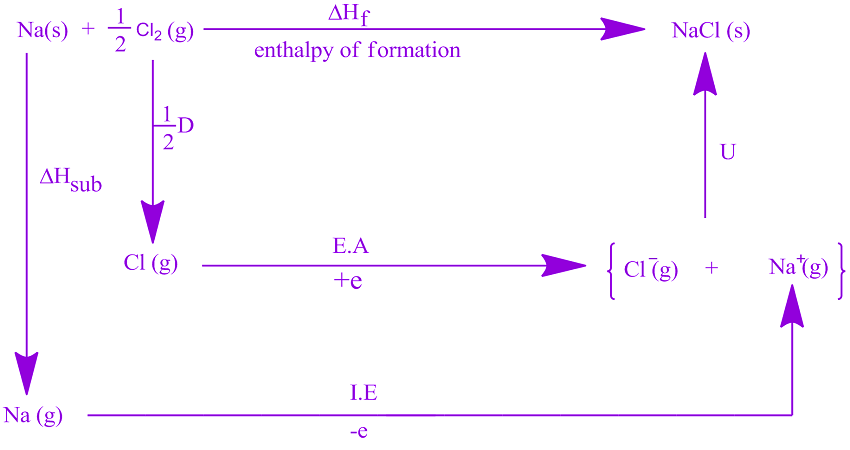
In general, ionization energy increases across the periodic table from left to right, and decreases from top to bottom. This process always requires an input of energy, and thus will always have a positive value.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
